What is the Difference Between Physical And Chemical Change?
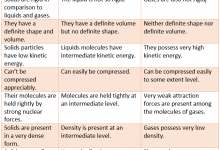
Difference Between Physical And Chemical Change is that physical change is a type of change in which the form of matter is altered but one substance is not transformed into another while chemical change is a process in which one or more substances are altered into one or more new and different substances.
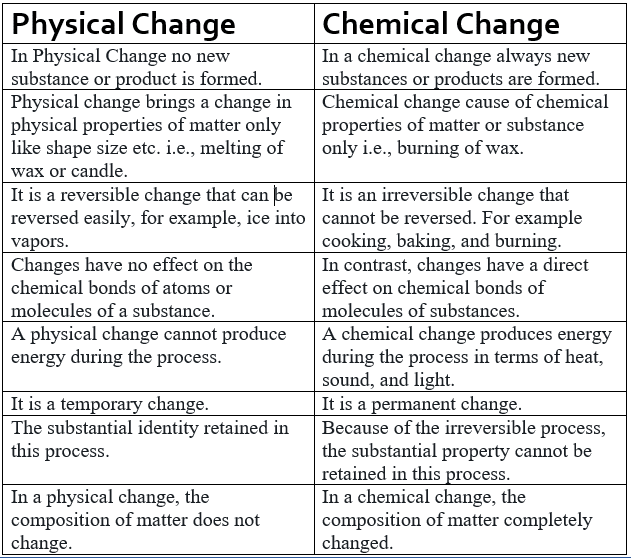
Difference Between Physical And Chemical Change in Tabular Form
| Physical Change | Chemical Change |
| in Physical Change no new substance or product is formed. | in a chemical change always new substances or products are formed. |
| physical change brings a change in physical properties of matter only like shape size etc. i.e, melting of wax or candle. | chemical change cause of chemical properties of matter or substance only i.e, burning of wax. |
| it is a reversible change that can be reversed easily, for example, ice into vapors. | it is an irreversible change that cannot be reversed. for example cooking, baking, and burning. |
| changes have no effect on the chemical bonds of atoms or molecules of a substance. | in contrast, changes have a direct effect on the chemical bonds of molecules of substances. |
| A physical change cannot produce energy during the process. | A chemical change produces energy during the process in terms of heat, sound, and light. |
| it is a temporary change. | it is a permanent change. |
| the substantial identity retained in this process. | because of the irreversible process, the substantial property cannot be retained in this process. |
| in a physical change, the composition of matter does not change. | in a chemical change, the composition of matter completely changed. |
What is a Physical Change?
A physical change transforms completely the composition of matter or substances but not the chemical composition. it is often used to separate mixtures into their components.
it is a contrary process than that of chemical changes in which the chemical composition of matter or substances are completely changed to make more or new substances in the end. the famous example of physical change is solving salt into water or melting of ice into water vapors.

it is a reversible process for example ice can be converted into water and water can be freeze into ice at very low temperatures. the physical change affects the color, size, shape volume, and density of the substances.
it can also change the strength of a given matter. physical changes are often taken into action to make changes in the arrangement of the elements to fulfill our necessity in daily life. they are important to make crystals.
10 Examples of Physical Changes in Our Daily life
we can see many more examples of physical changes in everyday life. it participates in building our houses to cooking. mixing sugar into water to make juices. ten common applications of physical change are given below:
- Mixtures of different substances
- Crystallization
- Heating and Cooling
- magnetism
- Aloys
- Breaking of a glass
- Chopping wood
- Boiling water.
- Crushing a can.
- Mixing sand and water
What is a Chemical Change?
A chemical change is altering the chemical composition of matter or substance when they are combined to make new substances or products. this term refers to chemical synthesis and usually occurs at least two or more substances during chemical reactions.
it is not a reversible change that completely changes the properties of reactant elements by producing new products.

the chemical reaction results in two types of reactions. those reactions which require heat to initialize or proceed are called exothermic reactions while the others that may have need heat to enable the process are called endothermic reactions.
you can find further detail about the difference between endothermic reactions and exothermic reactions. in this reaction, the product reactants are always had different properties to their participant substances.
10 Examples of Chemical Changes in our daily life
likewise the physical changes, chemical changes are also taking part in our daily life. we can see many examples in our surroundings like the burning of coal or wood. mixing cement with sand etc. some applications of chemical change are:
- Digestion of food
- Cooking an egg
- Baking a cake
- Rusting of iron
- Fireworks exploding
- Fermentation
- Burning of wood
- Metabolism
- Electroplating
- Change in color of fall leaves
hope that the above discussion will be enough to understand the difference between a physical change and a chemical change.
let us try to discover some common terms and questions that often confuse people about chemical and physical changes in the matter.
Is odor a physical or chemical property?
color, odor, melting point, boiling point, density, solubility polarity, and many other likewise terms are referred to be the physical properties.
Is burning wood a chemical change?
yes, the burning of wood is a chemical change because it is an irreversible process that can not be changed into the original reactant taking part in the reaction.
Characteristics of Physical Change
- During a physical change, only physical properties of substances change such as size, shape, color, and state.
- no very small amount of energy either evolved or absorbed.
- they are always reversible reactions.
- no new substances are formed.
- temporary reactions in nature
Characteristics of Chemical change
- in a chemical change, always new substances are formed.
- changes colors, smell, energy, the formation of energy or light.
You May Also Like: