What is the Difference between Solid Liquid and Gas?
The Basic Difference between Solid Liquid and Gas is that molecules are held together in solids by strong intermolecular forces of attraction, in liquids the attractive forces are at an intermediate level on attraction while in gas, the attractive forces between the gas molecules are very weak.
Everything around us is considered a matter. the matte is basically classified into Solids liquids and Gases as three different states of matter. all the states of matter i.e., solids liquids, and gases have distinct properties.
You May Also Like: the States of Matter and Their Properties
Characteristics and Properties of Solids
The particles of solid substances are tightly packed with each other. these are arranged in an order. there are strong attractive forces between them. the particles vibrate only at their mean positions. they cannot be compressed easily. that is why solids have a fixed shape and volume.

Characteristics and Properties of Liquids
the particles of liquid substances are very close to each other but are not arranged in an order. the attractive forces between them are strong but weaker than solids. liquids cannot be compressed easily because their particles are quite close to each other.
their particles can move away and towards each other and thus liquids can flow. liquids have fixed volume but their shape is not fixed. the liquids take the shape of the vessel in which they are kept.

Characteristics and Properties of Gases
the force of attraction between the particles of gases is very small. the distances between the gas particles are much greater than those of solids and liquids. gas particles or molecules move freely in all directions. they occupy all the available space.

they constantly collide with each other and with the walls of the container. in this way they exert pressure. gases have no fixed shape and no fixed volume. the liquids and solids have some distinct properties along with some similarities. for example, both have a definite volume.
the gases totally have changed in their properties. they don’t either have definite shape or volume. their molecules can move freely in all directions.
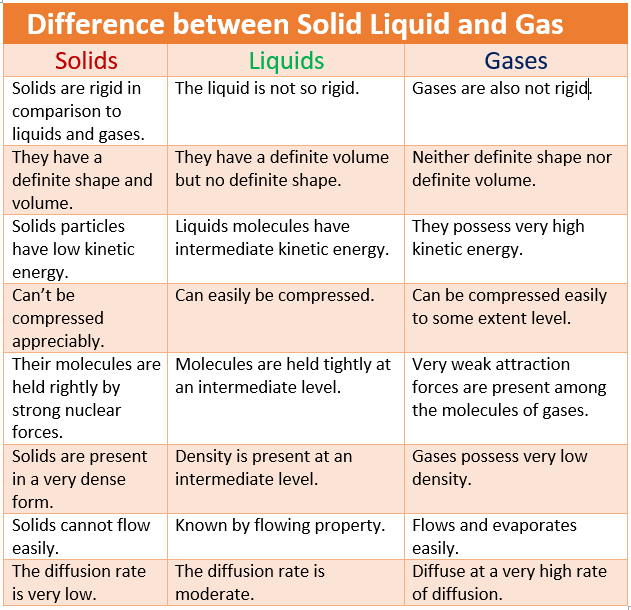
| Solids | Liquids | Gases |
| solids are rigid in comparison to liquids and gases. | The liquid is not so rigid. | Gases are also not rigid. |
| they have a definite shape and volume. | they have a definite volume but no definite shape. | neither definite shape nor definite volume. |
| solids particles have low kinetic energy. | liquids molecules have intermediate kinetic energy. | they possess very high kinetic energy. |
| can’t be compressed appreciably. | can easily be compressed. | can be compressed easily to some extent level. |
| their molecules are held rightly by strong nuclear forces. | molecules are held tightly at an intermediate level. | very weak attraction forces are present among the molecules of gases. |
| solids are present in a very dense form. | density is present at an intermediate level. | gases possess very low density. |
| solids cannot flow easily. | known by flowing property. | flows and evaporates easily. |
| the diffusion rate is very low. | the diffusion rate is moderate. | diffuse at a very high rate of diffusion. |
| very strong | less strong | weak |