what is the difference between organic and inorganic chemistry
organic vs inorganic chemistry
When the term chemistry is mentioned, many of us imagine these laboratories and the interactions in which materials are mixed and other materials are produced, but the chemistry is more profound.
The major difference between organic and inorganic chemistry is that organic chemistry is the study of the structure, composition, properties, reactions, and preparation of carbon-containing compounds including hydrocarbons while Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds.
[wp_ad_camp_1]
What is organic chemistry?
Chemistry divides into more than one section, one of which is called organic chemistry. Which contain carbon and hydrogen and how it behaves with other elements such as oxygen, silicon, nitrogen, sulfur, and phosphorus, for the ability to control the chemical reactions in terms of composition and color
it also determines the structure of the composite resulting from the reaction, Medical consciousness, and the production of natural materials and to produce new materials or improving existing materials before, and can also integrate organic chemistry in the metal industry so-called metal-organic chemistry, which is studying compounds containing carbon linked with other metals.
What is Inorganic chemistry?
Inorganic chemistry is a branch of chemistry that deals with the properties and behavior of inorganic compounds. Inorganic compounds are generally not biological and are characterized as containing no hydrogen.
[wp_ad_camp_2]
Organic chemistry is the study of any chemical reaction that involves carbon, an element upon which all life forms depend. The term “organic” is usually referred to as an animal or plant, so there is no misconception that organic chemistry always refers to biological processes.
This assumption is inaccurate. Many chemical processes deviate away from this line of thinking, and there are many biochemical processes that rely on inorganic chemical processes. There are exceptions to each rule. Although carbon is the main constituent of organic chemistry, inorganic chemicals also contain carbon.
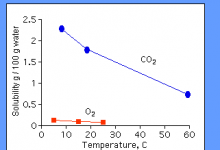
For example, carbon monoxide and carbon dioxide contain carbon but are inorganic compounds. Carbon dioxide is also very important for the chemical processes necessary for life, especially plant life. In fact, the lines between organic and inorganic chemistry are often unclear.
Difference between organic and Inorganic chemistry in the tabular form
[wp_ad_camp_3]
| ORGANIC CHEMISTRY | INORGANIC CHEMISTRY |
| Organic chemistry deals with organic compounds made of hydrocarbons. | Inorganic chemistry deals with inorganic compounds, typically those that have an ionic base. |
| Includes reactions that depend on the functional group present on the compound. | Includes acid-base reactions, displacement reactions, redox Reactions etc. |
| Mainly deals with salts and crystals. | Deals with oils, Fats, sugars etc. |
| Have higher melting points and degrade instead of boiling. | Melt and boil easily. |
External Links….!
http://www.softschools.com/difference/organic_versus_inorganic_compounds/157/