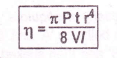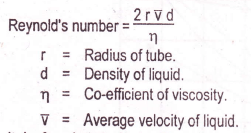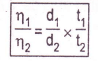Viscosity Definition and Formula with Units
Viscosity is a common observation that thick liquids like honey and mobile oil, flow slowly than many organic liquids like benzene and toluene, etc. It means that different rates of flow of liquids depend upon a certain property and that is viscosity.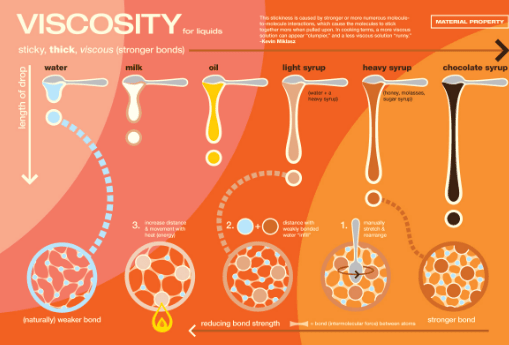
It is the property that opposes the relative motion of adjacent layers. In order to understand the viscosity, let us consider the flow of a liquid.
The liquid is considered to be arranged in a large number of parallel layers. The layer adjacent to the walls of the tube is stationary.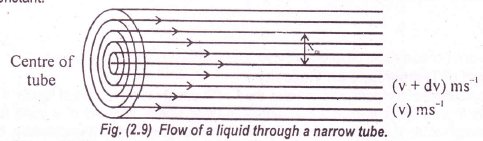
When we move towards the center of the tube, then the velocities of the successive layers go on increasing.
When the steady flow is reached then the velocity difference between any two layers becomes constant.
The force which is required to maintain the steady flow of liquid in direction of the force is directly proportional to the velocity gradient which is normal to the direction of flow. It is also proportional to the area of the contact ‘A’ between the moving layers of the liquids.
In the C.G.S system, the unit of “ŋ” is expressed as g cm-1 sec-1 and it is called Poise.
Definition and Units
“It is the force per unit area, needed to maintain a unit difference of velocity between two parallel layers of the liquid, unit distance apart.”
The units of viscosity ”ŋ” can be derived from the equation (1)
Unit of viscosity “Poise”
“When a force of one dyne per square cm is maintained, between two layers which are 1cm apart and the difference of velocity between the two layers is 1 cm per second, then it is called poise”.
1 centipoise = 10 -2 poise
1 millipoise = 10 -3 poise
Fluidity
“Fluidity is the reciprocal of viscosity.”
Ф = 1 / ŋ
The units of fluidity are reciprocal of viscosity i.e. (Poise)-1.
9 Examples of Viscosity in Everyday Life?
- pancake syrup
- honey
- ketchup
- engine oil
- cooking oil
- toothpaste
- lotion and cold cream
- Greece
- paints
Try Also: What are Examples and Applications of Viscosity in Everyday Life?
Effect of Temperature
The increase in temperature increases the kinetic energy of molecules of liquids. It means that a liquid starts flowing faster at high temperatures. In other words, a viscosity of a liquid falls with the increase in temperature. It is estimated that for each one-degree rise in temperature, there is a 2% decrease in viscosity.
Mathematical Relationship between Viscosity and Temperature
Arrhenius has given the following relationship between viscosity and temperature.
ŋ= Ae B/RT
A and B are the constants depending upon the nature of the liquid. According to this equation, an increase of temperature decreases the viscosities of liquids.
If we want to determine the constant ‘A’ and ‘B’ then we can convert it into the equation of a straight line by taking the natural log i.e. log to the base e. (It can be written as In)
If we plot a graph between 1/T on the x-axis and loge 17 (in ŋ) on the y-axis, then a straight line is obtained. From the slope of the straight-line ‘B’ can be calculated and from the intercept of the straight line factor, ‘A’ can be calculated.
The following graphs show that many liquids show straight lines. The anomalous behavior of H2O is seen from the graph because its hydrogen-bonded structure is broken with the rise in temperature.
temperature.
The quantity ‘B’ is the activation of energy for- the viscous flow. It is the energy barrier that must be overcome before the flow can occur.
It means that a molecule of a liquid should gain sufficient energy to push aside the surrounding molecules before it can take part in the liquid flow.
Coefficients of Viscosity of Liquids at 20°C in Poise and in SI Units (kg m-1 sec-1)
Remember that 1 pouse = 10-1 kg m-1s-1.
| Liquid | Viscosity (C.G.S. units) poise | Viscosity (SI units) kg m-1 s-1 |
| Benzene | 0.00652 | 0.000652 |
| Carbon tetrachloride | 0.00969 | 0.000969 |
| Ethanol | 0.01200 | 0.001200 |
| Ether | 0.00233 | 0.000233 |
| Glycerin | 14.9 | 1.49 |
| Water | 0.0101 | 0.00104 |
Try Also: Types of Viscosity
Measurement of Viscosity
General Principle
The measurement of viscosity of the liquid is based on Pioseulle’s equation (4)
Where,
V = Volume of liquid.
r = Radius of the capillary tube
I = Length of the tube.
t = Time of flow in seconds.
P = Pressure applied.
We should keep in mind that, equation (4) applies to the liquids when the rate of flow is slow and steady. The flow should be streamlined. Moreover, the diameter of the tube should be very small. If the flow rate is higher and the diameter of the tube is large, then the flow becomes turbulent.
In order to decide, which type of flow is there, we should use the empirical equation called Reynold’s number. It is the dimensionless quantity and its formula is as follows.
r = Radius of the tube.
d = Density of liquid.
ŋ= Co-efficient of viscosity.
V = Average velocity of the liquid.
It is found that if Reynold’s number is equal to or less than 2000, then the flow is streamlined. If the value is greater than 4000, then the flow is turbulent.
Measurement of Absolute Viscosity
It is difficult to measure directly the value of the absolute viscosity of a liquid using equation (1). The reason is that the measurement of ‘P’, ‘r’, and ‘V’ is difficult, so the viscosities of liquids are expressed in relative terms. “This is the ratio of the viscosity of the liquid to the viscosity of water taken as the reference standard and this is called relative viscosity.
“Ostwald’s viscometer to measure the relative viscosity of a liquid is a U-shaped glass tube with two marks ‘x’ and ‘y’. It has two bulbs as ‘A’ and ‘B’. The bulb ‘A’ is at a higher level than ‘B ‘. A definite volume of liquid is put in bulb ‘B’ and then sucked in bulb ‘A’. The time of flow of the liquid through the capillary tube is noted
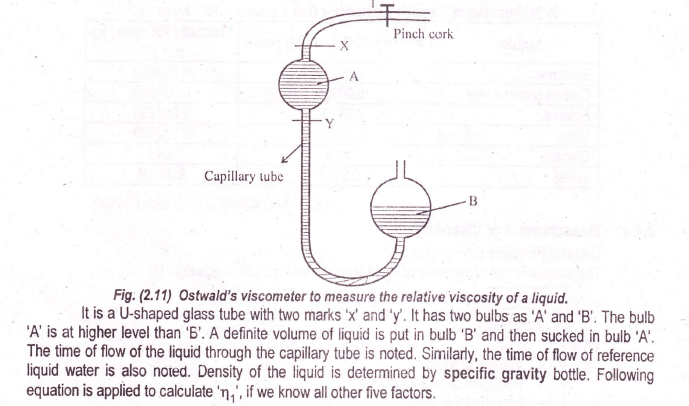 Similarly, the time of flow of reference liquid water is also noted. The density of the liquid is determined by a specific gravity bottle. The following equation is applied to calculate “ŋ1” if we know all the other five factors.
Similarly, the time of flow of reference liquid water is also noted. The density of the liquid is determined by a specific gravity bottle. The following equation is applied to calculate “ŋ1” if we know all the other five factors.
ŋ1 = viscosity of liquid
ŋ2 = viscosity of reference liquid
d1= density of the liquid, determined by specific gravity bottle
d2= density of reference liquid taken from the literature
t1= time of flow of liquid
t2= time of flow of reference liquid.
Viscosity and Constitution
Viscosity surely depends upon the structure of the compound. The following various aspects are very important to be considered.
- In the homologous series of organic compounds, the members differ from each other by- CH2 group. It has been observed that there is a gradual increase in viscosity values for the members of the homologous series.
- Some of the liquids are associated and so their viscosities Dunston in 1909, gave a useful relationship.
For the associated liquids, the values come out to very higher than 60. If d, M, and ŋ are measured in C.G.S. units, the following equation is used. The following Table shows that water, glycol, and glycerol are associated with liquids.
The following Table shows that water, glycol, and glycerol are associated with liquids.
| Liquid | (d/M) X ŋ X 106 | Conclusion |
| Acetone | 43 | Unassociated ” ” Associated More associated Highly associated |
| Toluene | 56 | |
| Benzene | 73 | |
| Water | 559 | |
| Glycol | 2750 | |
| Glycerol | 116400 |
It also depends upon the shape of the molecules. If the chain length of the molecule is shorter, the viscosity is less. The compounds of normal chain length have usually greater viscosity values than branched-chain Similarly, the viscosities of Tran’s isomers are greater than cis-isomers.
- The strength of intermolecular forces can also be depicted from the Values of viscosity.
- Water has a greater viscosity than ethyl alcohol due to greater hydrogen bonding in
- Just like the parachor of liquid, a parameter called Rheochor is also an additive and constitutive property. It depends upon the viscosity of the liquid.
[R] = M/d x ŋ1/8
We can verify the structure of the compound by comparing the theoretical and experimental Rheochors Table.
Measurement of Molar Mass of Polymer
The molar masses of polymers are very high. It depends upon the reaction conditions whether the molar mass of the polymer is high or low. The measurement of the viscosity of solutions of polymers can help us to find the molar masses of polymers.
Following relationship is used for this purpose
[ŋ]int = KMa
[ŋ]int = Intrinsic viscosity
M = Molar mass of the polymer
K and a = Constants, depending upon the nature of the solvent and the polymer
a = Factor depending upon the shape of the molecule.
For example, for random coiled molecules,
a =0.5
For rod-like molecules
a = 2
For spherical coiled molecules
a = 0
Intrinsic viscosity is a limiting viscosity number when the concentration of the polymer solution approaches zero.
ŋ sp = ŋ – ŋ0 / ŋ0
ŋ = Viscosity of a solution of the polymer in a solvent
ŋ0= Viscosity of pure solvent
A graph is plotted between concentrations of various solutions on the x-axis and ŋsp/C on the y axis. A straight line is obtained. This graph is extrapolated to the limiting value when c 0. The value of 11sp/C having the value at that stage is called ŋintrinsic.