Chemistry
What Is The Difference Between Molecule And Compound?
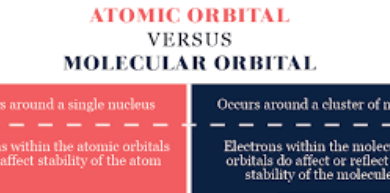
The Basic Difference Between Molecule And Compound is that A molecule is a group of two or more atoms bonded together by a chemical bond e.g. ionic or covalent bond while A compound is known as chemical species and is formed when two or more atoms joined together chemically.
Difference Between Molecule And Compound in Tabular Form
| Molecule | Compound |
| molecules are the smallest units of compounds and retain the characteristics of the compound. | A compound contains atoms of different elements that are chemically combined together with a fixed ratio. |
| A molecule is the smallest particle of a substance that retains the always chemical properties of the substance. it is a group of like or unlike atoms held together with chemical forces. | A variety of compounds can be found. it is always represented by a formula. |
| a molecule can be homonuclear or heteronuclear. | compounds are only heteronuclear molecules. |
| molecules have two types of bonds either ionic bond or covalent bond. | compounds have three types of bonds i.e., ionic bonds, metallic bonds, and covalent bonds. |
| they have both types o atoms either different elements or the same elements. | always have different elements. |
| Examples of molecules are Oxygen, Hydrogen, Nitrogen, Zinc, etc. | Examples of Compounds are Zinc Chloride, sodium chloride, calcium carbonate, etc. |
| Not all molecules are compounds. | all the compounds are made up of molecules. |
You May Also Like: