Modern Periodic Table (long-form)
Why do we need the Modern Periodic Table?
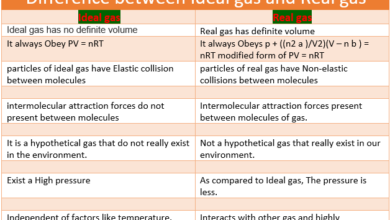
We need a Long form of Modern Periodic table of Elements due to the following reasons: As soon as a sufficiently large number of elements had been discovered and their properties well enough characterized, the need was felt to systematize them based on their properties. Attempts were made to classify the elements in a number of ways. The most noteworthy of these is that made by a Russian chemist, Dmitri Mendeleev, in, 1869 who gave a law known as Mendeleev’s Periodic Law which states as:
The physical and chemical properties of elements are a periodic function of their atomic masses. Mendeleev arranged the known elements in order of their increasing atomic masses in successive sequences so that the elements with similar properties fell in the same column in the form of a table which is known as Mendeleev’s Periodic table after his name.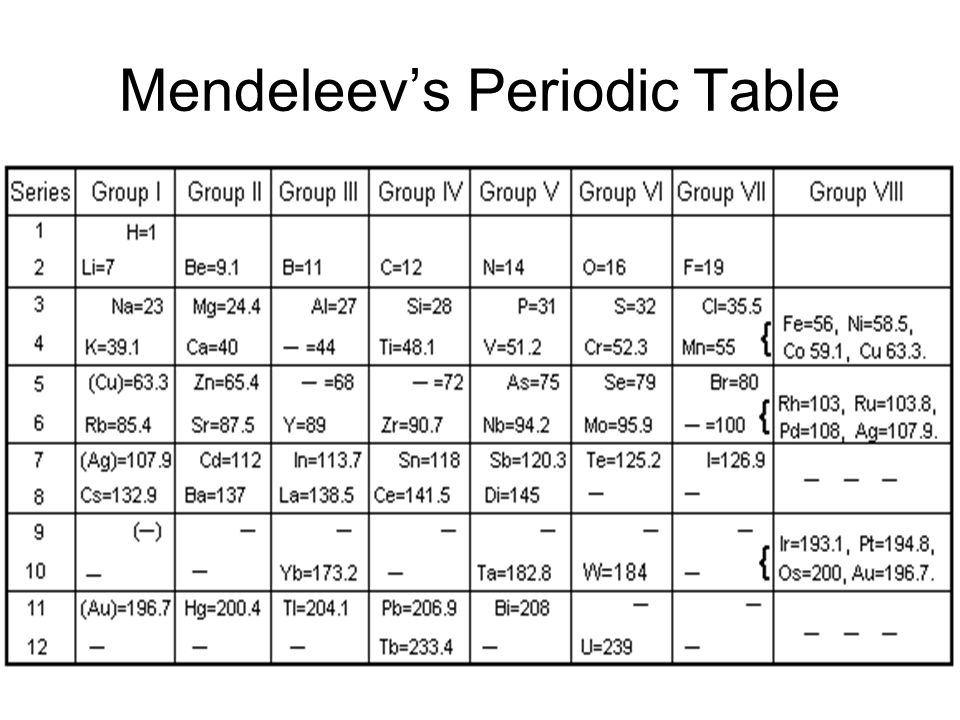
Mendeleev could not list the elements in order of increasing atomic number, as today’s periodic table does because atomic numbers were not known at that time. He did recognize that a listing of elements by atomic mass was not satisfactory. Mendeleev assumed that the observed chemical properties of the elements were more important than the atomic masses assigned to the elements-a good assumption: considering the suspect nature of many atomic masses of the time.
so, the question is :
Who arranged the modern periodic table?
In 1913 Henry Moseley developed the concept of atomic numbers. Moseley determined the frequencies of X rays emitted as different elements were bombard with high-energy electrons. He found that each element produces X rays of a unique frequency; further, he found that the frequency generally increased as the atomic mass increased.
He arranged the X-ray frequencies in order by assigning a unique whole number, called an atomic number, to each element. Moseley identified the atomic number with the number of protons in the nucleus of the atom and the number of electrons in the atom.’ The concept of atomic number clarified some problems in the early model of the periodic table which was based on atomic masses.
Modern Periodic Table (A Long form of Periodic Table)
The work of Moseley proved beyond doubt that the properties of elements are well explained and most of the anomalies and defects of Mendeleev’s Periodic Table disappear if the basis of the classification is changed from atomic masses to atomic numbers. This formulates the modern Periodic law which states,
The physical and chemical properties of elements are a periodic function of their atomic numbers. In order to remove the defects of Mendeleev’s periodic table a number of tables have been suggested for the classification of elements on the basis of modern periodic law. Out of such various tables, the one called long Form of Periodic Table is most widely used and is also referred to as Bohr’s Table.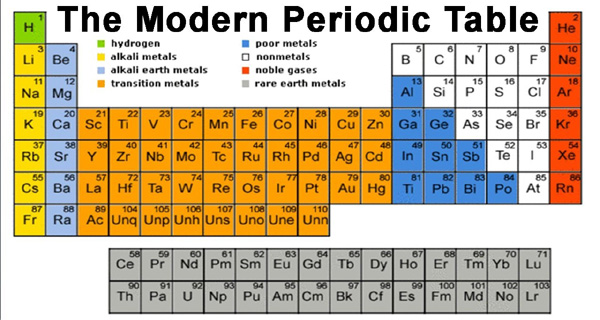
Since it is based on Bohr’s Scheme of the classification of the elements into four types based on their Electronic configurations. A tabular arrangement of elements in the order of increasing atomic numbers in rows and columns, highlighting the regular repetition of properties of the elements is called a periodic table. The basic structure of the periodic table is its division into rows and columns.
A modern version of the periodic table with the elements arranged by atomic number is shown in table 1.1. Each entry lists the’ atomic number, atomic .symbol, and atomic mass of an element. The horizontal rows in the periodic table are called periods. There are seven periods, which are numbered from 1 through 7 using Arabic numbers and they vary greatly in length.
Each period begins with an alkali metal and ends with an inert gas element. The vertical columns in the periodic table are called groups. There are 18 vertical columns which constitute 16 groups of elements. These are numbered 1A, IIA, IIIB ….. VIIB, VIIIB IB, lIB, IIIA—– VIIA and VIIIA (or zero) according to the traditional system of nomenclature (i.e. North American system of nomenclature). North America labels the groups with Roman numerals which have A and B designations.
Europeans use a similar convention that numbers the columns from IA to 8A and then from 1 through 18, thereby giving the label 7B (or VIIB) instead of 7A to the group headed by fluorine (F), and thus some columns have the A and B interchanging. To eliminate this confusion, the International Union of Pure and Applied Chemistry (IUPAC) suggested a new convention that numbers the groups from 1 through 18 with no A or B designations, as shown in table 1.1, which shows the traditional North American and the IUPAC conventions.
When we refer to an element by its periodic group, we will use the traditional North American convention. Elements in a group have similar chemical properties. The properties of elements in one group differ from the properties of elements in another group. Elements within a period have properties that change progressively across the table.
A period contains elements in order of increasing atomic number. Anew period is begun in order to place elements of similar properties in vertical columns. An element can be located in the periodic table by listing its period and group number Periods. These are the horizontal rows.
The first period (n=1) has two elements, hydrogen and helium, n indicates the number of main energy level for the elements in a period. Thus, the first period has only one main energy level for the elements.
The second period (n=2) has 8 elements, beginning with Li and ending with neon (Ne), i.e., Li3 — Ne10. The third period (n=3) also has 8 elements, beginning with Na and ending with Ar, i.e., Na11 to Ar18.
The first, second, and third periods are called short periods because they contain a small number (2 or 8) of elements.
The fourth and fifth periods consist of 18 elements each (K19 to Kr36) and (Rb37 to Xe54) respectively. Out of these, eight are typical (normal) elements which belong to A groups while the other 10 elements belong to B groups (except IIB) in the periodic table are transition metals because each of these elements either has an incompleted-subshell or readily give rise to cations that have an incompleted-subshell The sixth period has 32 elements Cs to Rn.
Out of these, eight in the A groups are typical elements, the elements in the B groups (except IIB; i.e., Hg) are transition metals, and fourteen from 58Ce through 71Lu are inner transition elements.
The inner transition elements also called the lanthanides or rare earth metals, are listed separately at the bottom of the table.
The fourth, fifth, and sixth periods are called long periods because they contain a large number (18 or. 32) of elements.
The seventh period is incomplete but is believed to be a long one (potentially 32 elements). The 14 elements 90Th through 103Lr, extracted from the seventh period and placed at the bottom of the table are called actinides.
At present this period has 23 elements which are from 87Fr to 109Une. All these elements are radioactive.
Out of these the elements namely 87 Fr, 88Pa, 89 Ac, 90Th, 91Pa, and 92U are 4 naturally occurring radioactive elements while the remaining elements (i.e., 93Np to 109Une are man-made (i.e., artificially prepared) radioactive elements and are called transuranium elements.
The reason for listing lanthanides and actinides separately is practical. If this were not done, the table would be too wide to fit on most pages, therefore, they are placed at the bottom of the periodic table to conserve space.
The separate listing also emphasizes the great similarities in the properties of elements in each of the two series.
Groups. There are 18 vertical columns of the periodic table which constitute 18 groups and are numbered as 1, 3, 4, 5,6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17 and 18 according to IUPAC recommendation adopted in 1984.
The elements belonging to groups 1, 2, 13, 14, 15, 16, 17, and 18 are known as normal or representative elements. Elements in-between groups 2 and 13 (i.e., 3-12) are called transition elements because they represent a transition (change) from electropositive elements on the left-hand side to electronegative elements on the right-hand side of the periodic table.
Two rows of elements (each. containing 14 elements) are known as lanthanides and actinides respectively. They are placed separately at the bottom of the periodic table to avoid uncontrolled sideways expansion of the periodic table.
According to the North American system, 18 vertical columns constitute 16 groups of elements. These are:
- IA, IIA, IIIA, IVA, VA, VIA, VIIA and VIIIA (zero groups) groups. The element of these groups is called normal elements. In other words, the A group elements in the periodic table are called normal or representative elements.
- IB, IIB, IIIB, IVB, VB, VIB, VIIB and VIIIB groups. Group VIIIB has three columns. IB, IVB, VB, VIB, VIIB, and VIIIB group have purely transition elements while in IIIB group some elements are transition elements and the remaining are inner transition elements. In other words, the elements in the B groups (except IIB) are known as transition elements.
It should be noted that there are three families grouped together under the group VIIIB because all the elements of group VIIIB have similar properties. These are separately referred to as iron, cobalt, and nickel families.
Elements with the same group number, but with different designations, have relatively few similar properties. Several groups of representative elements have common names.
The group IA elements (except H) are called the alkali metals, and the group IIA are called the alkaline earth metals. The group VIA are called halogens, the group VIA are called halogens and the group VIIA are called noble (or inert) gases.
Merits of Modern Periodic Table
The modern periodic table has a number of merits over the Mendeleev’s periodic table in the following respects:
- The arrangement of the elements is based on a more fundamental property namely atomic number.
- In this periodic table, the position of an element is related to the electronic configuration of its atom.
- It explains the similarities and variations in the properties of the elements in terms of electronic configurations and brings out clearly the trends in chemical properties across the long periods.
- The inert gases have completely filled electron shells that have been placed at the end of each period. Such a location of the inert gases represents a logical completion of each period.
- The greatest advantage of this periodic table is that this can be divided into four block elements namely s-, p-, d- and f-block elements.
- It provides a clear demarcation of different types of elements like active metals, transition metals, non-metals, metalloids, inert gases, lanthanides, and actinides.
- Due to the separation of the two sub-groups, dissimilar elements (e.g.) alkali metals and coinage metals) do not fall together.
- The lanthanides and actinides have been given a separate place at the bottom of the periodic table due to their analogous chemical behavior.
- This arrangement of the elements is easy to remember and to reproduce.
Defects of Modern Periodic Table
Although the modern periodic table has many advantages, it has some defects are given below:
- The problem of the position of hydrogen still remains unsolved.
- This periodic table is unable to include lanthanides and actinides in its main body.
- this periodic table does not reflect the exact distribution of electrons among all, the elements.
- Chemical helium is an inert gas but since the remaining members of the inert gas group possess the electronic configurations ns2p6 against that of He —> 1s2, a different place should be given to helium.