Passive and Active Transport Models with Mechanisms
Mineral Transport Models: Movement of molecules and ions between different compartments in a biological system is called transport. It is a highly selective process and during transport, some compounds accumulate against the concentration gradient while others are not transported at all.
Transport between the cell and its environment is controlled by the plasma membrane the separates cytoplasm from the external environment determines the type of molecules that move into and out of the cell and direction and rate of transport.
[wp_ad_camp_1]
However, in most plants the vacuole occupies up to 90% of the cell’s volume and contains the bulk of the cell solutes, therefore tonoplast also serves as selective barrier and transport between cytosol and vacuoles is controlled by it.
Passive and Active Transport Models
The ion concentrations in the cytosol and vacuole are controlled by passive and active transport processes, therefore the transport of a particular ion or polar molecules may be active transport or passive transport.
Passive Transport and Passive Transport Mechanisms
The diffusion of a substance across a biological membrane s called passive transport because the cell does not have to spend energy in the transport. Numerous investigators demonstrated the absorption of ion without the expenditure of cellular energy.
They found that when a plant cell or tissue is transferred from a dilute salt solution to a concentrated salt solution, the tissue absorbs ions rapidly due to the concentration gradient it is not affected by temperature or metabolic inhibitor showing that metabolic energy is not involved.
How ions accumulate against chemical potential gradient without the expenditure of metabolic energy has been explained by several forms of absorption such as facilitated diffusion. Donnan effect and equilibrium, ion exchange and mass flow of ions.
[wp_ad_camp_2]
I. Facilitated Diffusion
It is found that diffusion of ions does not take place through lipid bilayers as water does and these are transported with the help of various transport proteins called membrane transporters that span across the membrane. This phenomenon is called facilitated diffusion. The transport is towards the direction of the lowest concentration.
A transport protein has many of the properties of an enzyme. Just an enzyme is specific for its substance; a membrane protein is specialized for the solute it transports and ha specific binding site similar to the active site of the enzyme. However, a membrane protein may also transport a family of related substances.
For example, in plants, the K+ transporter on the plasma membrane may transport Rb+ and Na in addition to K but K+ is preferred. Unlike enzymes, the transport proteins do not catalyze the chemical reactions. When a solute molecule binds with transport protein, a change in the shape occurs. 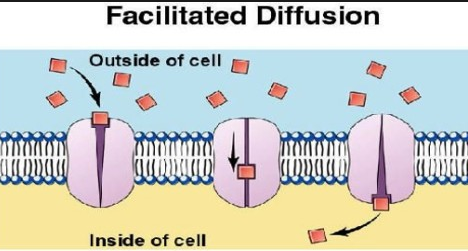
There are two types of membrane transporters. Channels and carriers, that acts as a door to the cells.
[wp_ad_camp_3]
- The channels are proteins that function as pores in the membranes. They are highly selective and facilitate the passage of specific molecules in either direction. The size of a pore and the density of surface charges on the interior lining determine its transport specificity.
- The carriers are proteins with an active site to which the transport substances are bound. The bounding results in a change in the shape of the protein, which exposes the substance to the solution on the other side of the membrane where the substance dissociates itself from the carrier’s binding site.
Facilitated diffusion has the following essential characteristics:
- It is specific, with only certain molecules able to pass a given channel.
- It is passive; the direction of the net movement is determined by the relative concentration of the transported molecule inside and outside the membrane.
- It may become saturated if all the protein channels are in use.
- It provides a cell with a ready means or preventing buildup of unwanted molecules within the cell.
However, diffusion cannot account for accumulation against a concentration gradient. Many studies indicate that solute transport into cells is strongly dependent upon MT and the ability of the cells to respire in the presence of oxygen and produce ATP.
[wp_ad_camp_4]
ii. Donnan Effect and Equilibrium
Membrane Potential
All cells have voltages across their plasma membranes. The cytoplasm of the cell is negative in charge compared to the extracellular fluid because of an unequal distribution of anions and cations on opposite sides of the membranes. The voltage across a membrane is called a membrane potential and it ranges from -50 to -200 millivolts.
The membrane potential acts like a battery, an energy source that affects the traffic of all charged substances across the membrane. Because the inside of the cell is negative compared to outside, the membrane potential favors the passive transport of cations into the cells and anions out of the cell.
Thus, two forces drive the diffusion of ions across a membrane, a chemical force (the ion concentration gradient), and an electrical force (the effect of the membrane potential on the ion’s movement).
This combination of forces acting on an ion is called the electrochemical gradient. Therefore, an ion does not simply diffuse down its concentration gradient but diffuses down its electrochemical gradient.
Non-Diffusible or Fixed Anions
Proteins behave like ions because the R-groups of their amino acids may bear positive or negative charges. Most soluble proteins behave like anions because they possess more negative than positive sites. The proteins being larger in size are unable to cross the membranes. Therefore, the protein anions are non-diffusible or fixed. The presence of these fixed anions inside the cells affects the absorption of ions.
[wp_ad_camp_5]
J W. Gibbs and F. G. Doman described the effect of these fixed anions on the equilibrium distributions of small ions. This is called the Donnan effect and equilibrium. The Donnan effect does not influence the equilibrium distributions of non-ionized substances such as glucose, etc.
Model for Dorman Effect and Equilibrium
Let us consider a membrane having fixed anions on its inner side. However, it is- freely permeable to cations and anions in the external solution. Equal numbers of cations and anions from the external solution will diffuse across the membrane until equilibrium is established.
Normally, this equilibrium would also be electrically balanced, but the presence of fixed anions on the inner side of the membrane imbalance the electrical equilibrium. Therefore, additional cations are required t6 balance the negative charges, so cations will move into the cell from an external solution.
This results in an increase of cation concentration in the internal solution. Also because of fixed anions, the concentration of anions in internal solution will be less than anions in the external solution. So anions will more in.
[wp_ad_camp_1]
When the product of anions and cations in the internal solution is equal to that of anions and cations in the external solution, the Donnan equilibrium is attained.
[Ci+] [Al–] = [C0+] [Ao–]
Where
Ci+ = cations inside
AI– = anions inside
Co+ = cations outside
Ao– = anions outside
Thus, the accumulation of ions against a concentration gradient can occur without the participation of metabolic energy until Dolman equilibrium is reached.
Donnan equilibrium serves as an explanation for the accumulation of ions against the concentration gradient without expenditure of energy.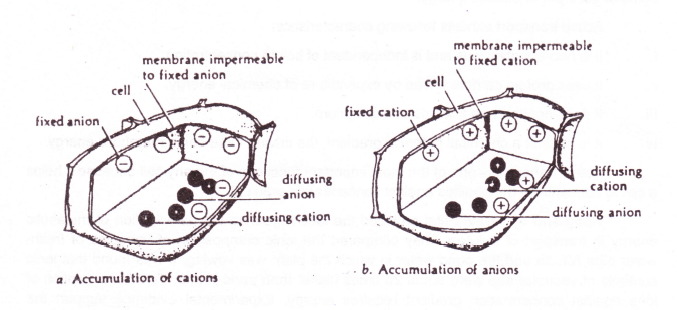
iii. Ion Exchange
Ions attached or adsorbed on the cation exchange surface present in the cell walls or membranes of tissue may exchange with ions from the external solution in which the tissue is immersed, in the same way as ion exchange takes place between the soil solution and ions adsorbed on the surfaces of colloids.
[wp_ad_camp_2]
For example, the cation K of the external solution is exchanged with H ion adsorbed to the surface of the membrane. The anions are possibly exchanged with the free hydroxyl ions (0H) in the same manner.
The ion exchange mechanism allows greater absorption of ions from the external solution than absorbed by free diffusion. Therefore, the ions can accumulate against the concentration gradient.
iv. Mass Flow of Ions
Some investigators believe that ions can move through roots along with the mass flow of water. According to this theory increase in transpiration should increase the rate of absorption of ions. The participation of transpiration may be direct, the transpiration pull causes mass flow of water and ions move with water from the soil solution; or indirect. Transpiration removes ions from xylem ducts resulting in a decrease of chemical potential so more absorption of ions takes place.
The theory is generally accepted. Lopushinsky in his experiments with tomato plants proved indirectly that increase in transpiration results in an increase in salt absorption. He applied different degrees of hydrostatic pressure to dropped to string plant enclose in pressure chambers containing nutrient solutions of radioactive P32 and Ca45. He showed that an increase in hydrostatic pressure brings about an increase in the amount of radioactive phosphate and r calcium moved into the xylem.
Active Transport and Active Transport Models
Active transport is my transport of an eon or solute across a membrane that requires some direct input of cellular energy.
[wp_ad_camp_3]
Active transport exhibits the following characteristics:
- It is non-spontaneous and is independent of solute concentration.
- It uses protein carriers driven by the expenditure of chemical energy.
- It does not proceed towards equilibrium.
- It is against a chemical potential gradient, the driving force being metabolic energy.
Active transport is one of the most important functions of any cell because it helps a cell to concentrate metabolites against a concentration gradient.
Hoagland and Davis first provided the idea regarding the participation of metabolic energy in the transport of solutes. They compared the ionic composition of vacuoles of freshwater alga Nitetta and the pond water in which the plant was vowing.
It was found that ionic contents of vacuolar sap were about 25 times higher than pond Water. This accumulation of ions against the concentration gradient requires energy. Experimental evidence supports the participation of metabolic energy in the active transport of ions. If cellular metabolism is reduced by using respiratory inhibitors, the ability of the cells to carry active transport is reduced.
[wp_ad_camp_4]
Many investigators suggested various concepts regarding the active uptake of ions. These include electrogenic transport, cotransport and ATPase transport.
Electrogenic Transport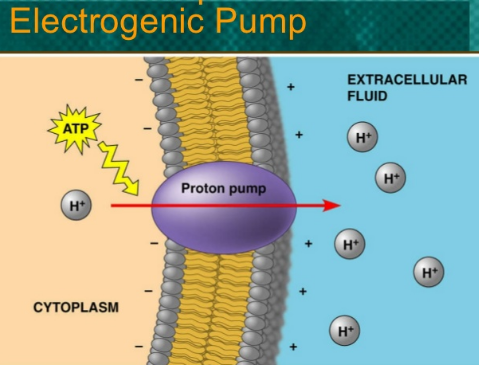
Some membrane proteins actively transport ions and contribute to the membrane potential. For example, the protons pump in plants which actively transport hydrogen ions (protons) out of the cell.
The pumping of H+ ions transfers a positive charge from the cytoplasm to the outside. A transport protein that generates voltage across a membrane is called an electrogenic pump. This carrier-mediated active transport is called electrogenic transport.
An electrogenic pump works as followings:
[wp_ad_camp_5]
- The carrier protein embedded in the membrane hinds the cation (M+) on the inside of the cell. It is phosphorylated by ATP.
- The binding results in a change in the shape of carrier protein (conformational change) that exposes the cation to the outside and makes it possible for the cation to diffuse away.
- The phosphate ion (P) from ATP is released to the inside of the cell.
- The release of phosphate ion restores the original shape of the carrier protein and allows the pumping of a new cycle again.
Proton Pump in Plants
Proton pumps are the main electrogenic pumps of plants. Using ATP for power, a proton pump translocates a positive charge in the form of hydrogen ions (H +). The proton pump generates membrane potential (voltage) and creates a proton gradient across a membrane.
This voltage and hydrogen gradient are used by the plant in uptake of sugar and other nutrients. When proton diffuses back across the membrane through special channels, the transport is coupled to the production of ATP within the cell (chemiosmosis).
A classic example of proton pumping is regulation of turgor in guard cells that regulates the stomatal apertures. It has been observed that protons are extruded into the apoplast during stomatal opening, and there are separate pumps for potassium and chloride into guard cells.
Examples of Passive Transport
- Dissolution in the phospholipid layer
- The release of enzymes and hormones into the bloodstream
- Entrance by integral protein channels
- Glucose absorption
- The renal glomeruli
- Gas diffusion
- The action of insulin
- The roots of the plants
- Sweating
- Intestinal absorption
Examples of Active Transport
- Sodium-potassium pump
- Phagocytosis
- Calcium pump
- Exocytosis
- Pinocytosis
- Transcytosis
- HIV infection
- Sugar phosphotransferase
- Iron catchment
- LDL uptake
You May Also Like This:


