Classification of Enzymes with Definition, Structure, and Characteristics
In this post, you are going to learn about the Classification of Enzymes step by step with Diagrams.
This post includes:
- An overview of Enzymes
- Various functions of the Enzymes
- Structure and Characteristics
- Lots more
So if you want to get benefits from this post you’ll love this post.
Let’s Dive right in..
The enzymes are the biological catalysts. the major Characteristics of Enzymes is that they the biologically active proteins that speed up chemical reactions in the cell but they don’t bring change in the reaction. being proteins, they are always folded into chains of amino acids with a specific shape and bonds.
OR
“Enzymes are the organic compounds composed of proteins, act as biocatalyst and increase the rate of a chemical reaction without being changed”.
Enzymes increase the efficiency of biological reactions. They are specific for each type of reaction. The reactions proceed at a very slow speed without enzymes. Therefore, life is impossible without enzymes.
Structure of Enzyme
They have the following components:
· Globular Structure
Enzymes are composed of hundreds of amino acids. These amino acids join together and form a globular structure.
· Active Site
The catalytic activity is restricted to a small portion of the enzyme called the active site. It is composed of a few amino acids. All other amino acids form the globular structure of the enzyme. This active site is involved in the catalytic activity. The reactants are called substrates. The substrates attach to the active site.
The active site of the enzymes is made up of two regions:
- Binding Site: It recognizes the specific substrate and forms the enzyme-substrate complex. This reaction activates the catalytic site.
- Catalytic Site: The activated catalytic site changes the substrate into products.
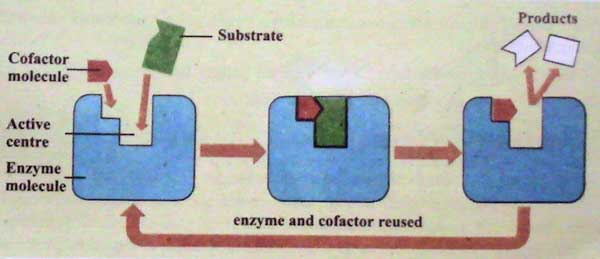
· Co-factors
Some enzymes contain a non-protein part, called co-factor. The co-factors are essential for the proper functioning of these enzymes. The cofactors are essential for the proper functioning of these enzymes.
The co-factors act as a bridge between the enzyme and the substrate. They often directly take part in the chemical reactions. Sometimes, the co-factors provide a source of chemical energy. This energy is necessary to start a reaction. There are three types of co-factors:
- Activator: The detachable co-factors are called activators. These co-factors are inorganic ions like Mg+2, Fe+2, Cu+2
- Prosthetic Group: The covalently bonded non- protein part cofactors are called prosthetic groups.
- Coenzyme: The loosely attached non-protein part with an enzyme is called a coenzyme. The coenzymes are organic but non-protein in nature. They are related to vitamins. The vitamins act as raw material for the synthesis of coenzymes. Coenzymes are not destroyed during a reaction. Thus, they can be used again and again. Therefore, a small amount of vitamins is needed in the cell.
· Apoenzymes
An enzyme with a coenzyme or prosthetic group removed is called apoenzymes. Apoenzymes remain inactive.
· Holoenzymes (Apoenzymes + Co-factors)
An activated enzyme, consisting of the polypeptide chain and cofactor is called Holoenzyme. A holoenzyme is formed when a cofactor attaches with the apoenzymes. It makes the enzyme activity and it starts the reaction.
Location of Enzymes
Many enzymes are simply dissolved in the cytoplasm. Some enzymes are tightly bound to certain sub-cellular organelles. The enzymes are produced in the cell. They are used near the site of their production. For Example;
- Enzymes of photosynthesis are found in the chloroplast.
- Some enzymes are involved in cellular respiration. These enzymes are present in mitochondria.
- The enzymes involved in protein synthesis are present in ribosomes.
Characteristics of Enzymes
Enzymes are biological catalysts. They have the following characteristics:
- All enzymes are globular proteins.
- They increase the rate of reactions. But they are not used in the reactions.
- They do not change the nature or properties of the end products.
- A small amount of enzyme can act on a large number of substrate molecules.
- Enzymes are very specific in action. A single enzyme catalyzes only a single substrate or group of related substrates.
- They are sensitive to change in PH, temperature, and substrate concentration.
- Some enzymes require a co-factor for their proper functioning.
- They lower the activation energy. The minimum amount of energy required to start a reaction is called activation energy.
- Some enzymes are potentially damaging if they become active in the wrong place. For example, pepsin is a protein-digesting enzyme. It can destroy the internal structure of cells. Thus, it is produced in an inactive form called pepsinogen. The enzymes are formed in the cells and are closed in lysosomes. The lysosomes are membranous bounded bodies.
- These are not consumed in the chemical process. After the process, these are separated off and can be used again.
- In some enzymes, a non-proteins part is also present, called a prosthetic group.
- Enzymes are used for specific chemical reactions. Each enzyme is not used for all reactions.
- They can react to both acidic and alkaline substances due to the presence of proteins.
- The activity of enzymes can be accelerated by certain ions or salts, called activators such as Ni, Mn, Mg, and Cl, etc. They decrease the amount of activational energy.
Mechanism of Enzyme Action (catalysis)
· Specificity of Enzymes
An enzyme is a three-dimensional globular protein. Each enzyme has a specific composition. It has specific amino acids and has a specific shape. Due to this specificity, each enzyme reacts with a specific substrate into the product. In the end, the enzyme releases without any change. So this enzyme can be used again and again.
· Chain of Enzymes
In some cases, enzymes act in series or chains. These enzymes are present in a specific order. Each enzyme controls a specific reaction. One enzyme hands over the substrate to another enzyme. In this way, they form an enzyme coenzyme chain. The product formed by the one enzyme is transferred to the next enzyme. Finally, the end-product is formed.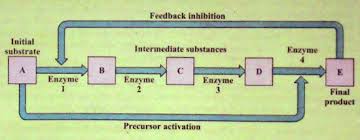
· Formation of the enzyme-substrate complex
An enzyme has an active site. The active site on an enzyme is a three-dimensional cavity. It contains specific charges. The enzyme reacts with the substrate by these charges.
The amino acids of the enzyme form the charge and shape of the active site. These amino acids come close to each other and form polypeptide chains. These polypeptide chains fold and coil. This folding and coiling form the globular structures of the enzyme. Thus, the amino acids are arranged in a definite way. The active site of the enzymes is made up of two regions.
1) Binding sites
It recognizes the specific substrate and forms ES (enzyme-substrate) complex. This reaction activates the catalytic site.
2) Catalytic Site
The activated catalytic site changes the substrate into products. The enzyme detaches from the products after the completion of the reaction. It remains unchanged during the reaction. The enzymes require an aqueous medium for their activities.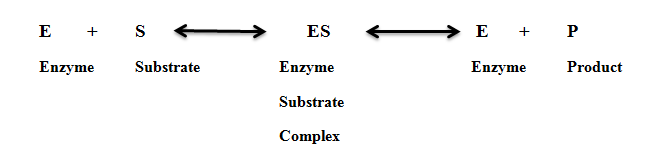
Mode of Action of Enzyme
The action of the enzyme depends upon the structure of the enzyme. It has three-dimensional structures. It has an active site, which is of a particular size, and shape. This active site will attach to substrate. The non-active site is termed as an allosteric site.
Two theories have been proposed about the action of Enzymes.
A.Lock and Key Theory
Fisher (1898) proposed this theory about the action of the enzyme. According to this theory, each enzyme can react with a specific substrate in the manner of a lock and key, just like a lock can be unlocked by a particular key.
The site of the enzyme, which is active and attached to the substrate, is called an active site. The other site is known is an allosteric state. This site helps in the catalytic process. Sometimes, other molecules are connected with active sites, but there is no bond formation and no chemical reaction takes place.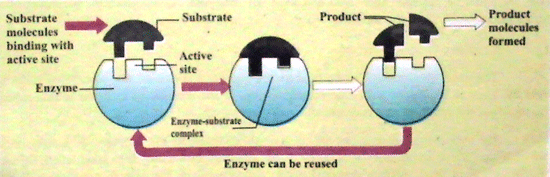
B.Induce Fit Model Theory
Koshland in 1959 proposed another theory about the action of an enzyme, called the Induced Fit Model Theory. According to this theory, when the enzyme combines with a substrate, some changes occur in the structure of the enzyme, due to this change, the enzyme performs its catalytic function in more effective performance.
Factors Affecting the Rate of Enzyme Action
The enzymes perform specific functions. This specificity of an enzyme is due to its specific chemistry and shape. Many factors affect the chemistry and shape of enzymes. Therefore, these factors also affect the rate of catalysis. The following factors affect the rate of enzyme action.
· Enzyme Concentration
If the amount of substrate is unlimited, then the rate of reaction directly depends on the concentration of the enzyme. The rate of reaction becomes double by the amount of the enzyme.
The increase in the number of enzyme molecules increases the number of active sites. Therefore, more active sites change more substrates into products. After some time, the amount of substrate becomes limited. Now, the reaction rate cannot be increased with the increase in enzyme concentration.
· Substrate Concentration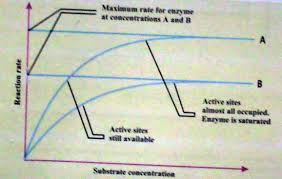
At a low concentration of substrate, the rate of reaction is directly proportional to the amount of substrate. If the enzyme concentration is kept constant and the amount of substrate is increased, a point comes when a further increase in the amount of substrate does not increase the rate of reaction.
The substrate molecules occupy all the active sites of the enzymes at the high concentration of the substrate. Thus, a further increase in the substrate concentration does not increase the reaction rate.
· Temperature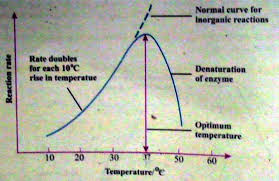
The rate of reaction may increase with the increase in temperature. But this increase is limited. Every enzyme has an optimum temperature. The specific temperature at which an enzyme can work at maximum rate is called optimum temperature. For example, 370C is the optimum temperature for the enzymes of the human body.
Heat provides two types of energies:
1. Activation Energy:
Activation energy accelerates the chemical reactions at high temperatures.
2. Kinetic Energy:
Heat also provides the kinetic energy to the molecules. Therefore, the reactant molecules move quickly. Thus the chances of their collision with each other are increased and they easily react with each other.
[wp_ad_camp_3]
The high amount of heat energy also increases the vibrations of atoms of the enzymes molecules. Further increase in vibration can destroy the globular structure of the enzymes. This globular structure is essential for the activity of the enzymes. It ultimately denatures the enzyme.
· PH Value
The antilog or negative log of the Hydrogen Ion concentration is called PH. Every enzyme has an optimum PH value. An optimum PH is a narrow range of PH in which an enzyme functions effectively.
A slight change in PH can change the ionization of the substrate. This change in ionization reduces or blocks the activity of the enzyme. Extreme changes in PH break the bonds of the enzyme. It may cause the denaturation of the enzyme.



Hey There. I found your blog using msn. This is an extremely neatly written article. I will be sure to bookmark it and come back to learn extra of your useful information. Thanks for the post. I will certainly return.
Great web site you have got here.. It’s difficult to find quality
writing like yours nowadays. I really appreciate people like you!
Take care!!
Thanks for the excellent article