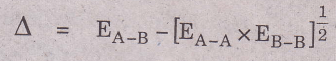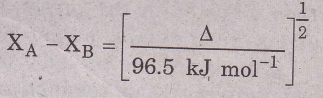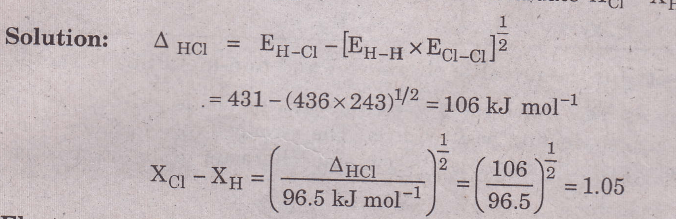Electronegativity: Scale and Definition with Examples & Trends in Periodic Table
The relative tendency of a bonded atom in a molecule to attract the shared electron pair towards itself is called electronegativity. In other words, electronegativity is a measure of the ability of an atom in a molecule to draw bonding electrons to itself. The greater an atom’s electronegativity, the greater is its ability to attract electrons to itself.
It is an inherently fundamental property of the atom and is fundamentally different from electron affinity. Since electron affinity represents the tendency of an isolated gaseous atom to attract the electrons while it represents the tendency of a bonded atom to attract the shared electron pair.
The electronegativity of an atom in a molecule is related to its ionization energy and electron affinity, which are properties of isolated atoms.
It depends on the structure of the atom and the number and kind of atoms with which it may combine. Generally, small atoms are more electronegative. The atoms with nearly filled shells of electrons have a higher electronegativity than those with less than half-filled or scarcely filled.
For example, halogens are the elements with nearly filled shells and hence they are the most electronegative, while alkali metals are the elements with scarcely filled and are least electronegative.
Factors Affecting the Magnitude of Electronegativity
Its magnitude depends on the following factors.
(i) Atomic Size
The smaller the size of an atom, the greater is its tendency to attract towards itself the shared pair of electrons. Thus smaller atoms have greater electronegative values than the larger atoms.
(ii) The number of Inner Shells
The atom with the greater number of inner shells has less value of electronegativity than the atom with a smaller number of inner shells, e.g., the electronegativity values of halogens decrease from F to iodine since the number of inner shells increases from F to I.
(iii) Charge on the Ion (i.e., Oxidation State)
Electronegativity is a variable quantity. It varies with the change in the 6xidation state of the element. The element in higher oxidation state has more value of electronegativity than in the lower oxidation state. Thus the value of electronegativity of Fe3+ is higher than that of Fe2+.
(iv) Type of Hybridization
The magnitude of electronegativity of an atom also depends on the type of hybridization which the atom undergoes in the formation of different bonds in the molecule. The magnitude of electronegativity increases as the s—character in hybrid orbitals increases.
(v) Ionization Energy and Electron Affinity
The atoms of the elements which have higher values of ionization energy and electron affinity also have higher values of electronegativity.
(vi) Nature of Atoms to which the Atom is Bonded
Since electronegativity of an atom is not the property of an isolated atom, it depends on the number and nature of the atoms to which the atom is bonded. Therefore the electronegativity value of an atom is not constant. For example, the electronegativity value of P atom in PCl3 the molecule is different from that in the PF5 molecule in which the number and nature of the atom both to which P atom is bonded change.
Periodic trends of Electronegativity
electronegativity increases from left to right in a period in the periodic table since nuclear charge increases and atomic radius decreases which cause electronegativity to increase across a period.
With some exceptions (especially within transition metals), electronegativity decreases in going down a group due to the successive appearance of inner shells and. increase in atomic radius wit an increase in atomic number.
The addition of extra Inner shells in larger atoms screen the shared pair from the nucleus and. thus the electron pair is attracted less by the atoms in the combined state. The values are Unitless.
Electronegativities of elements (Pauling’s Values)
| H 2.20 | |||||||||||
| Li 0.98 | Be 1.57 | B 2.01 | C 2.55 | N 3.04 | O 3.44 | F 3.98 | |||||
| Na 0.93 | Mg 1.31 | Al 1.61 | Si 1.90 | P 2.19 | S 2.58 | Cl 3.16 | |||||
| K 0.82 | Ca 1.00 | Sc 1.36 | Ti 1.54 | …… | Cu 1.90 | Zn 1.65 | Ga 1.81 | Ge 2.01 | As 2.18 | Se 2.55 | Br 2.96 |
| Rb 0.82 | Sr 0.95 | Y 1.22 | Zr 1.33 | …… | Ag 1.93 | Cd 1.69 | In 1.78 | Sn 1.96 | Sb 2.05 | Te 2.1 | I 2.66 |
| Cs 0.79 | Ba 0.89 |
Electronegativity Scales: Measurement of Electronegativity
A number of scales have been devised to measure the electronegativity of the atoms. These scales are arbitrary and are based on various types of experimental data like bond energy, dipole moment, ionization potential, and electron affinity.
More commonly used scales are described below:
Mulliken’s Scale: In 1934 R.S. Mulliken suggested on theoretical grounds that the electronegativity (X) of an atom be given as half it’s ionization energy (IE) minus electrons affinity (EA).
X=IE-EA/2
Until recently, only a few electron affinities had been measured. For this reason, Mulliken’s scale had, limited utility.
Pauling’s Scale:
This scale is based on an empirical relation between the energy of a bond (called bond energy) and the electronegatjrjj5 of the bonded atoms. Pauling defined electronegativity difference for two dissimilar atoms A and B of a molecule A—B, in terms deviation () of the A—B bond energy from the geometric mean of A—A and B—B bond energies.
Pauling suggested that if two atoms A and B have the same electronegativity, the bond energy of the A—B bond would be equal to the geometric mean of A—A and B—B bond energies. This would be so when the electrons in the bond would be equally shared in purely covalent bonds in all the three cases.
Pauling Scale Image Formula
Pauling observed that for the majority of A—B bonds the energy exceeds the geometric average because generally, the atoms A and B have different electronegativities and there is an ionic contribution to the bond in addition to the covalent one
Let the difference between A—B bond energy and the geometric mean of A-A and B-B be Δ, then
This excess energy is known as the ionic resonance energy.
Pauling proposed that this excess energy, Δ can be used as an empirical basis to determine. electronegativity difference. If XA and XB represent the electronegativities of A and B respectively, the difference XA — XB could be related to Δ as.
The factor 96.5 kJ mol converts Δ from kJ mol-1 (SI units) to electron volts (eV) per molecule.
Example: The bond energies of H2, Cl2 and HCl are EH_H 436kJ mol-1;
Ecl-cl = 243 kJ mol-1; EHCI = 431 kJ mol-1. Evaluate Xcl — XH.
Electronegativity and Nature of the Chemical Bond
The emphasis on atoms in molecules indicates the electronegativity is a bond property and not a property of an isolated atom. Electronegativity values help in deciding the nature of a bond in a molecule.
Elements with large differences in electronegativity (a metal and a nonmetal) tend to react with each other to form an ionic compound. Elements with low electronegativity differences (two nonmetals) tend to form covalent bonds. There is an arbitrary rule to distinguish between bond types.
When the two bonded atoms have an electronegativity difference of 0.5 or less, we shall call the bond nonpolar. In polar bonds, the electronegativity difference is between 0.5 and 1.7. In the ionic bond, the difference is greater than 3.7.
Orbital Electronegativity
electronegativity is believed to depend on the nature of orbital involved in bonding. s-orbital penetrates nearer to the nucleus than an orbital, therefore s-orbital contributes more electronegative character. The orbital electronegativities, therefore, depend on the percentage of s and p character in sp3,sp2, and sp hybridization.
The magnitude of electronegativity increases as the s-character in hybrid orbital increases. Thus for a carbon atom, the electronegativities (on Pauling scale) are 2.48, 2.75, and 3.29 in sp3,sp2 and sp hybridization, respectively.
The commonly used electronegativity value of 2.5 for carbon is based on sp3 hybridization.
Group Electronegativity
The electronegativity of an atom adjusted for the presence of substituent is called group electronegativity. The group electronegativity of CH3 or CC13 will be the electronegativity of carbon adjusted in the presence of three H or Cl atoms.
Several methods making use of kinetic data, atomic electronegativities, and other physical measurements, have been developed to calculate the group electronegativities. Electronegativities of some common groups are listed below:
Electronegative Values of some Groups
| Group | Electronegativities | Group | Electronegativities |
| CH3 | 2.3 | CN | 3.3 |
| CF3 | 3.35 | COOH | 2.85 |
| CCl3 | 3.00 | C6H5 | 3.0 |
Electropositivity is the converse of electronegativity. It is related to the tendency to lose electrons yielding positive ions. The stronger the tendency of an element to lose electrons, the more is its metallic character or electropositive character.
M → M+ + e-
From a chemical standpoint, metal atoms display a tendency to lose one or more electrons when entering into compound formation with nonmetal atoms, The nonmetal atoms in these combinations show a tendency to gain one or more electrons.
The tendency to lose electrons depends on the ionization energy, thus electropositivity is evidently linked with ionization energy of the element.
Metallic Nature
The smaller the ionization energy of the element, the greater will be its metallic character. Since it is easier to remove an electron from a larger atom than from a smaller one, the metallic character increases as we move down from the top to bottom in a given group of the periodic table.
Since the atomic size decreases and ionization energy increases as we move across a period, the metallic character decreases from left to right across a period in the periodic table. When two atoms have the same size, the one with the larger atomic number would be less metallic in nature.
The general increase of metallic character in going down a group is also confirmed by the fact that the oxides of the elements of a group become more and more basic as we proceed from top to bottom in a group.
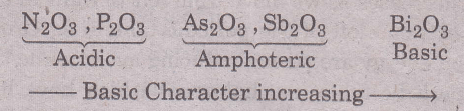
For example, the oxides of group VA elements (M203) become more and more basic from N203 to Bi203 as
shown below:
The general decrease of the metallic character of the elements in going from left to right in a period is also confirmed by the fact that the oxides of the elements of a given period become less and less basic or more or more acidic as we proceed from left to right as shown below for the oxides of elements of 3rd period.
- Na2O (Strong basic)
- MgO(Basic)
- Al2O3(Amphoteric)
- SiO2 (feebly Acidic)
- P2O5(Acidic)
- SO3(More acidic)
- Cl2O7(Most Acidic)
- Acidic Character increasing →
Non-Metallic Character
The greater is the value of electron affinity of an element, greater is the tendency to accept the electron to give the anion. This means that greater is the value of electron affinity of an element, greater is its non-metallic character or electronegative character.
Since electron affinity values decrease from top to bottom in a group, the non-metallic character of the elements also decreases from top to bottom in a group.
Similarly with the increase of electron affinity values from left to right in a period non-metallic character of the elements also increases.
The trend of variation shows that s- and p-block elements (i.e., normal elements) lying at the lower left-hand side of the periodic table show maximum electropositive character and hence are metals, while the elements lying at the upper right-hand side show minimum electropositive character and hence are nonmetals, The elements lying in between metals and non-metals show characteristics of both metals and non-metals and hence are called metalloids.