Effective Nuclear Charge in Periodic Table with General Formula
Effective Nuclear Charge is the net positive charge of attracting the electron. The effective nuclear charges, Zeff, equals the number of protons in the nucleus, Z, minus the average number of electrons, S, that are between the nucleus and the electron in question:
Zeff=ZS
Thus, the positive charge experienced by outer-shell electrons is always less than the full nuclear charges because the inner-shell electrons partly offset the positive charge of the nucleus. The inner electrons are said to shield or screen the outer electrons from the full charge of the nucleus. This effect is called the screening effect.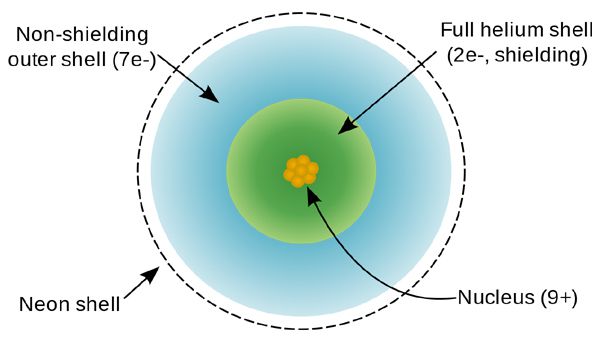
The effective nuclear charge is the positive charge that an electron experiences from the nucleus, equal to the nuclear charge but reduced by any shielding or screening from any intervening electron distribution.