Structure of chlorophyll With Formula & Meaning, Definition
(What does chlorophyll do to your body?)
Chlorophyll is a green pigment. It is present in all photosynthetic organisms. It is insoluble in water but it is soluble in organic solvents like carbon tetra chloride (CCl4), alcohol, etc.
Types of chlorophyll
There are different kinds of chlorophyll. These are chlorophyll a, b, c, and d. These pigments are found in eukaryotic photosynthetic plants. The chlorophylls present in photosynthetic bacteria are called bacteriochlorophylls.
Wavelength absorbed by chlorophylls
Chlorophylls absorb mainly violet-blue and orange-red wavelengths. Chlorophylls least absorb green and yellow wavelengths. So, these wavelengths are transmitted or reflected. The dark green color always masks the yellow color. Hence plant appears green. It remains green unless some other pigment masks it.
Structure of chlorophylls
A chlorophyll molecule has two main parts:
Hydrophilic Head
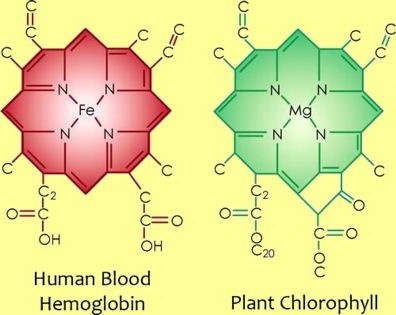
It is flat, square, and light absorbing part. The Head has a complex porphyrin ring. This ring is made up of four jointed smaller units called pyrrole rings. Each pyrrole ring is composed of carbon and nitrogen atoms. An atom of magnesium is present in the center of the porphyrin ring.
This Mg atom coordinates with the nitrogen of each pyrrole ring. That is why the deficiency of magnesium makes the plant yellow. The Haem group of the hemoglobin is also a porphyrin ring. But it contains an iron atom in place of magnesium in the center.
Hydrophobic hydrocarbon tail
It is a long and anchoring part. It is composed of a long hydrocarbon chain called Phytol (C20OH39). This Phytol is attached to one of the pyrrole rings. The chlorophyll molecule is embedded in the hydrophobic core of the thylakoid membrane with this tail.