Electron Configuration and the Modern Periodic Table
How is the electron configuration related to the periodic table?
The electron configurations of elements are related to their location in the periodic table. The periodic table is structured so that elements with the same pattern of outer-shell (valence shell) electron configuration are arranged in columns.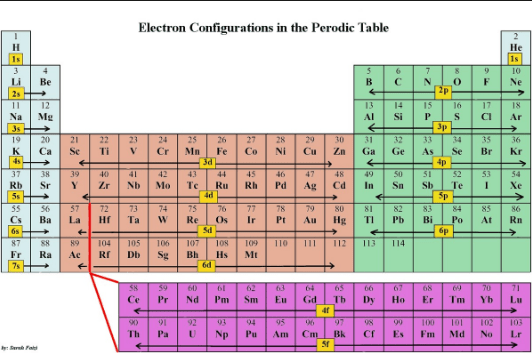
For example,
the group lIA elements all have ns2 outer electron configurations, while the group lllA elements have ns2, np1 configurations.
[wp_ad_camp_1]
The total number of orbitals in each shell is equal to n2: 1, 4, 9 or 16. Because each orbital can have 2 electrons, so each shell can accommodate up to 2n2 electrons: 2, 8, 18 or 32.
The structure of modern periodic table reflects this orbital structure.
The first row has two elements (H and He elements), the second and third rows have eight elements, the fourth and fifth rows have 18 elements, and the sixth row has 32 elements (including the lanthanide metals).
Some of the numbers repeat because we reach the end of a row of the periodic table before a shell completely fills.
For example,
the third row (period) has 8 elements, which corresponded to filling the 3s and 3p orbitals. The remaining orbitals of the third shell, the 3d orbitals, do not begin to fill until the fourth row (period) of the periodic table (and after the 4s orbital) is filled. Likewise, the 4d orbitals do not begin to fill until the fifth row of the table, and the 4f orbitals do not begin, filling until the sixth row.
[wp_ad_camp_2]
All of these observations are evident in the structure of the periodic table. For this reason, the periodic table is our best guide to the order in which the orbitals are filled.
One can easily write the electron configuration of an element based on its location in the periodic table.
Notice that the elements can be grouped in terms of the type of orbitals into which the electrons are placed.
On the left are two columns of elements. These elements, known as the alkali metals (group IA is1) and alkaline earth metals (group IIA is 2) are those in which the outer-shell s orbitals are being filled. On the right is a block of six columns.
These are the elements of group lllA (ns2np’), group IVA (ns2np2) group VA (ns2np3), group VIA (ns2p4), group VIlA (halogens, ns2np5), group VIIl A (the noble gases, ns2np6), in which the outermost p orbitals are being filled.
The s-block and the p block of the periodic table contain the representative (or main-group) elements.
In the middle of the table is a block often columns that contain the transition metals.
These are elements in which the d orbitals are being filled. Below the main portion of the table are two rows, each row contains 14columns.
These elements are often referred to as the f-block metals because they are the ones in which the f.orbitals are being filled.
[wp_ad_camp_3]
Recall that the numbers 2,6,10 and 14 are precisely the number of electrons that can fill the s, p, d and f subshells, respectively.
A close inspection of the periodic table shows that the electron configurations of certain elements appear to violate the rules. For example, the electron configuration of Cr is [Ar] 4s13d5 rather than [Ar]3s23d4, as we might have expected.
Similarly, the configuration of Cu is [Ar] 4s1 3d10 instead of [Ar] 4s23d9. The anomalous behaviour is largely a consequence of the closeness of the 3d and 4s orbital energies.
It frequently occurs when there are enough electrons to lead to precisely half-filled sets of degenerate orbitals (as in Cr) or to a completely filled d subshell (as in Cu). – There are a few similar cases among the heavier transition metals (those with partially filled 4d or 5d orbitals) and among the f-block metals


