Composition of Soil Factors affecting and Water Contents
Soil is the medium of growth for plants and the plants are anchored in the soil, through their roots. Similarly, large amounts of water are continuously required by a plant during its life cycle. The water is largely absorbed from the soil by plants through their roots. Therefore, for a better understanding of the absorption of water the composition of soil and the anatomy of roots must be taken into account.
Composition of Soil
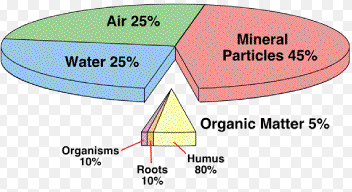
Soil is a heterogeneous mixture of inorganic mineral particles, decaying organic matter, and living microorganisms, along with air and various inorganic salts and organic molecules dissolved in water (soil water).
1. Inorganic Mineral Particles
The mineral particles are present as sand, smaller silt, and still smaller clay particles. The sand particles exist as coarse sand and fine sand. In addition to these gravels are also found. These soil particles are known as soil fractions or separate.
The soil particles are classified on the basis of their sizes. A soil has a specific distribution of particle sizes. Soil texture refers to the size of particles that make soil.
Practically all soils are mixtures of sand, silt, and day. The soils with about lo to 25’’o clay and rest about equal parts of sand and silt are called foams. If the percentage of sand is more the loam is called sandy loam and if more clay particles are present than the normal ratio the loam is called clay loam.
The soil with more clay particles is called clay soils. The clay particles are colloidal in nature and can hold most water and there is less air space between the particles.
Such soil lacks the oxygen necessary for root respiration. Similarly, root penetration is restricted in clay soils. But the clay particles provide sites for the adsorption of ions present in the soil solution.
The silt particles are non-colloidal and do not absorb water but act as capillaries in soil. The water is held among these capillaries and becomes available to the roots of the plants for absorption. The sand particles are porous; therefore the roots can penetrate easily in Soils containing sand particles.
The sandy loam and clay loam are considered to be the best soils for the growth of plants and agricultural purposes. These soils hold adequate water, have sufficient air spaces, and are easily penetrated by roots.
ii. Organic Matter
The remains of plants, animals, and microorganisms are acted upon by the chemicals present in the soils and also by the soil microorganisms and break organic matter in the soil.
This results in the release of inorganic ions and C02 and colloidal organic residues are left. This is called humus and the process of formation of humus from dead organic matter is called humification.
Humus consists largely of dark-brown organic molecules rich in phenolic compounds derived mainly from the lignin of plant residues. The humic particles mix intimately in soil and form the matrix of the soil.
The humic particles are colloidal particles that become hydrated with water and provide adsorption sites for mineral ions present in the soil solution. These ions become available to plant roots for absorption. Further degradation of humic particles results in the formation of minerals in a process called mineralization.
iii. Soil Water
Water is the second-largest component of the soil. Most inorganic chemical compounds and organic molecules present in soil are soluble in water; therefore the soil water is termed as soil solution.
The water in the soil exists in various states such as:
- Any water that moves downward through moist soil in response to gravity is called gravitational water. It moves down below the reach of plant roots, therefore unavailable for absorption. The water in the deep and permanently saturated zone is called groundwater, and. the upper surface of this layer is called the water table.
- Capillary Water: If we examine the soil a few hours after watering or rain, the upper layer of soil is dry and the only water left in the soil is that remaining n capillary pores. This water held in the capillaries is called capillary water. It is the source of almost all the water available to plant roots for absorption.
- Hygroscopic Water: Water molecules are also found around the colloidal particles (clay or humic particles) as hygroscopic water. Plants can absorb only a relatively small amount of hygroscopic water.
- Chemically Combined Water: Some of the water is also present chemically’ combined with the chemical compounds present in the soil, for example, hydrated oxides of aluminum, iron, silicon, etc.
Water Contents of the Soil
- Field Capacity: The percentage of moisture when the drainage of a wetted soil slows down is called its field capacity. The moisture in the soil at field capacity usually lies between 5% and 40%. The field capacity indicates the point at which the force of capillarity (cohesion) is equal to forces of adhesion. Field capacity is also a measure of the storage capacity of the soil.
- Water Contents of the Soil: The weight of water in a given weight of soil in an oven at 105 ‘C is called the water content of the soil. It is expressed in terms of the percentage of dry weight. For example, ¡f a sample weighs 60 gm. when moist and only 40 gm. when oven-dried, the moisture content is 20/40 x 100 = 50%.
- Permanent Wilting Percentage: The amount of capillary water is reduced when it is removed from the soil by evaporation and absorption by plant roots. The water supply to plant cells becomes insufficient and the cells lose turgor with the result that plant wilts.
- The phenomenon is termed wilting. The earliest wilting may be evident only during the hottest part of the day, but finally, it becomes so complete that wilted plants will not revive even though their foliage is brought to 100% relative humidity and transpiration is stopped. This condition is called permanent wilting.
The percentage of water that remains in the soil when the permanent wilting is attained is called the permanent wilting percentage or wilting coefficient. It varies from 1 and 15% depending upon the texture of the soil. Soil scientists have agreed to consider soil with a water potential of -1.5 MPa to be at the permanent wilting percentage.
4. Soil Atmosphere: The gases confined to the soil pores in a given soil are referred to as soil atmosphere or soil air. The main gases constituting the soil atmosphere are oxygen, carbon dioxide, and nitrogen. The carbon dioxide contents of soil air are several hundred times, higher than the normal carbon dioxide concentration in the atmosphere.
The content and composition of soil air are determined by the water content of the soil since air occupies those soil pores not filled with water. Therefore, poorly drained soils are poor ¡n air contents thus affecting the growth of plants and soil microbes.
5. Soil Organisms: Soil accommodates a diverse population of diverse populations of living organisms, both animals and plants. These range in size from rodents and large tree roots to worms, insects, and soil fungi and bacteria, these organisms influence the physical, chemical, and biological properties of soil.
Insects and earthworms help in the physical breakdown of plant residues while fungi and bacteria decompose the dead organic matter and form humus. The decomposition of dead organic matter also results in the release of essential nutrients.
FAQs (Frequently Asked Questions)
What are the 5 components of soil?
Answer: the soil is composed of mixtures of different components. the major 5 components of soil are water, air or gases, minerals, organic compounds, and microorganisms.
What is soil composition and properties?
Answer: the composition of soil includes the ratio of air or gases, minerals, organic compounds, and microorganisms, etc. Textures, structure, porosity, chemistry, and color are called properties of soil.
What is normal soil?
Answer: if A soil having a profile less or more in equilibrium with our environment is known as normal soil.
What are 2 main ingredients of soil?
Answer: soil is basically divided into two major ingredients i.e., Abiotic and Biotic ingredients. the Abiotic ingredients consist of air, minerals, plants, and all those Non-living things while biotic factors include all living things.
What are the three main layers of soil?
Answer: the layers of soil are divided into three horizons which are as follows:
- surface horizon
- subsoil
- substratum
You May Also Like:
